Can We Appoint Our EU Medical Device Distributor as Our Importer?
No doubt you landed here because you did some research and figured out that you now need an EU-based regulatory importer. Perhaps you have been exporting medical devices to Europe for many years and are wondering if your EU medical device distributor can also fulfill the role of importer for you.
Let’s clarify one thing straight away. “Importing” can involve three different processes:
- Physical handling and movement of devices
- Customs activities
- Placing the device on the market
Role of a Regulatory Importer
Before we address the importer role, we want to share that the need for an importer is not new. Section 3.3 of the Blue Guide has defined the importer’s role for many years. Article 13 of the European Medical Device Regulation now expressly requires an importer with specific obligations for any medical devices being imported into the European Union.
MedEnvoy can act as your independent EU importer. Request a proposal.
Four Things to Consider when Using your Distributor as your EU Importer
So, back to your question. The answer is “yes” – your EU Medical Device Distributor(s) can technically act as your importer. You can also have multiple importers, for example, in different countries. But there are several considerations to make before you ask a distributor to act as your EU importer.
Your EU Medical Device Distributor May Not Want to Be Your Importer
You should not assume that your EU Medical Device Distributor will want to serve as your importer. While the importer is primarily a “behind the scenes” operator, they have many responsibilities as the entity responsible for placing your devices on the market and are financially liable if non-compliant devices are placed on the EU market. Distributors are not.
Fines Are Possible if Obligations Are Not Met
The fines levied by each EU country can be significant if these obligations are not correctly followed, particularly if these have led to non-compliance with the MDR. Distributors may not be up to the task of fulfilling those obligations.
Sharing Documentations with Your EU Medical Device Distributer
Because the responsibilities of the importer and their associated liability are significant, they will need to have access to and carefully examine your technical documentation to ensure compliance with the MDR. Sharing technical documentation with your distributor as your importer is perhaps not something you would like to do.
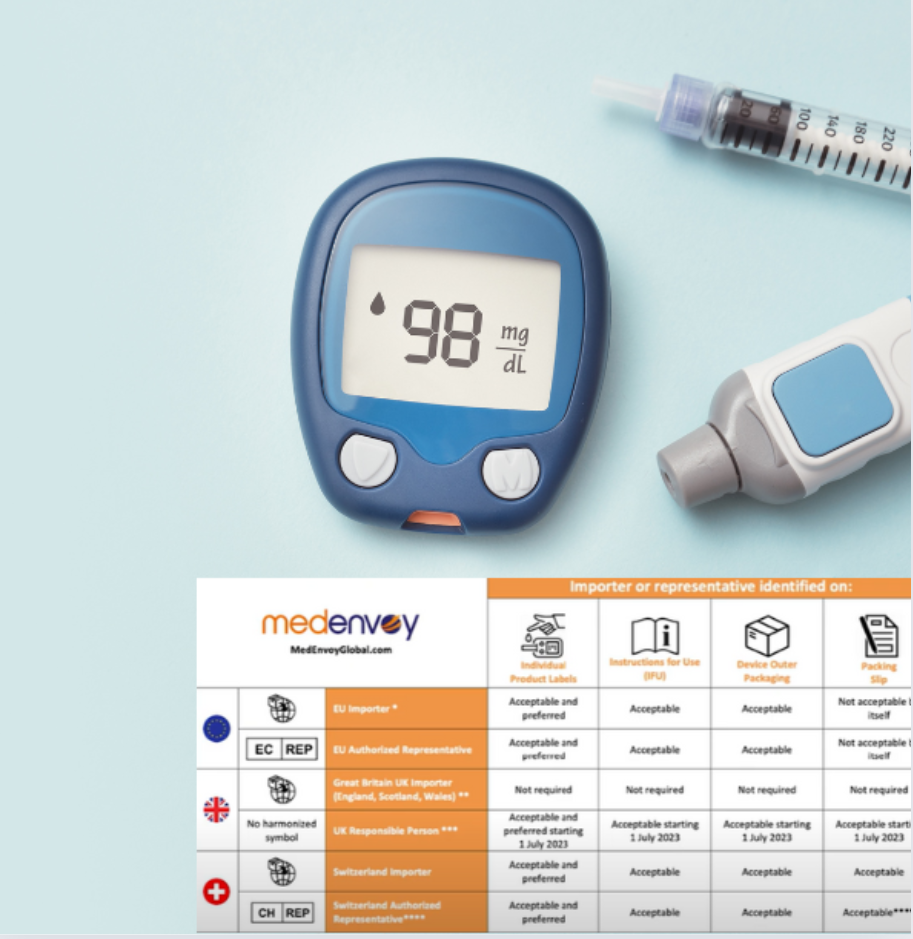
EU Medical Device Labeling Requirements
Your selected importer needs to be clearly identified up to the point where the device is “put into service.” In practical terms, this means all the way to the end user. When you have multiple distributors, having one of them identified as the importer may not be commercially acceptable by other distributors.
Alternative to Selecting an EU Medical Device Distributor as Your Importer
Selecting an “independent” importer that is solely focused on executing on its importer obligations and that will ensure MDR compliance without a need to take physical possession of your devices, is an option MedEnvoy Global offers to non-EU based manufacturers as an alternative to selecting a distributor(s) as your importer.
In addition, if you do have an EU based legal entity, but do not have the resources or structure to fulfill the MDR obligations of the importer, MedEnvoy Global can perform all of the “back office” responsibilities of the importer while still allowing you to act as the official importer. This means your name (as importer) will appear on device labeling/packaging/IFU, as required by the MDR and IVDR, but MedEnvoy will conduct the tasks of the MDR importer on your behalf.